Zirconia puzzle: what makes zirconia unique and how to choose the right zirconia
Zirconia may be part of your everyday vocabulary, but how much do you know about this ceramic material? Learn more…

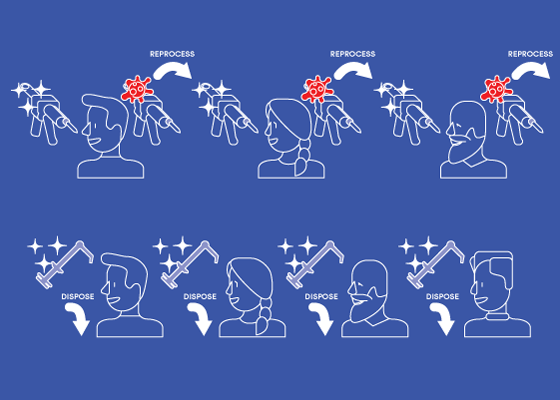
Should you reprocess and reuse, or pitch after each patient? Learn what to consider when choosing between single or multi-use products in a post-COVID environment.
Post-COVID, everything will change – from your approach to PPE and scheduling patients, to the procedures you perform. But should your products change too? One factor you may be considering is when to incorporate a single-use solution and when to stick with multi-use delivery. Let’s take a closer look at infection prevention, product choices and what you may choose to change in a post-COVID dental setting.
The CDC classifies product infection risk on a scale of non-critical to critical. Non-critical products pose the lowest risk, as they only encounter intact skin, which acts as a barrier to most microorganisms. Semi-critical products are those that are used intra-orally or on non-intact skin, increasing the risk of infection. Critical products are considered high risk, as they’re used to penetrate soft tissue or bone. The infection risk is lowest when it comes to non-critical products and highest when it comes to critical products.1
Nonetheless, products under each umbrella can be reprocessed safely. After use, non-critical items should be cleaned, and then disinfected if the product is visibly soiled. Guidelines indicate that heat-tolerant, semi-critical dental products should be heat sterilized. The CDC recommends that heat sensitive, semi-critical products should be reprocessed using high level disinfection or replaced with a disposable alternative. The FDA has published an additional guidance for multiple use intra-oral dental dispensers, such as those that deliver adhesive or composite materials.2 It states that these devices that cannot be sterilized or high level disinfected based upon their design must be covered with a barrier sleeve during use to prevent contact with mucous membranes. Multi-use critical products, on the other hand, must always be sterilized between patient uses.
According to the CDC, dental offices should consider adopting single-use products when they replace multi-use, semi-critical products that cannot tolerate heat sterilization.3 The guidelines also indicate that similarly classified, multi-use products may be used safely if the manufacturer’s reprocessing instructions are followed correctly.
But why should you consider single-use products and delivery systems? Single-use products take the risk of patient-to-patient infection out of the equation – infection control is baked into the product design. Of course, the risk of patient-to-patient transmission is also addressed when a multi-use device has been appropriately reprocessed. But if it gives you peace of mind, or if you think it may give your patients confidence, replacing multi-use products that are routinely used intraorally with single-use alternatives may be the right way to go.3
Further, the CDC advises that offices should consider not just the initial product cost of a single-use item, but also the time, cost and material savings obtained by eliminating the need for sterile processing of that product.3 At a time when so many procedures and protocols have ratcheted up and your office workflow is almost unrecognizable, every factor – from expenses to application needs to infection risk – is worth exploring.
Dental products, of course, are not one-size-fits all. Variables like application needs, ergonomic or control considerations, and more may well be worth reprocessing a multi-use version of a product rather than switching to a single-use option.
Zirconia may be part of your everyday vocabulary, but how much do you know about this ceramic material? Learn more…
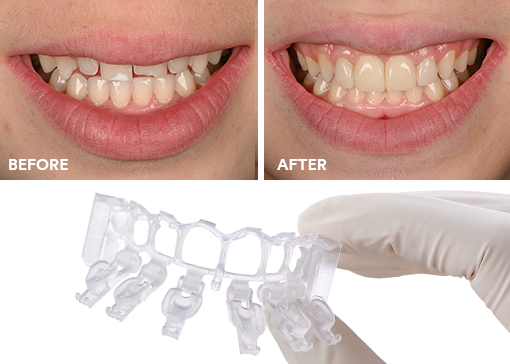
Achieving the correct shape is vital to a natural-looking anterior composite restoration. Learn how the 3M™ Filtek™ Matrix, paired with…
Direct composite restorative procedures can be challenging, particularly when it comes to esthetic cases. Discover how new techniques and tools…